This disproved Dalton’s claim that atoms are the smallest particles of matter. In short, Thomson had discovered the existence of particles smaller than atoms. He found that the mass of the particles was 2000 times smaller than the mass of the smallest atom, the hydrogen atom. He did this by determining how much the cathode rays were bent when he varied the voltage. Thomson also measured the mass of the particles he had identified. 
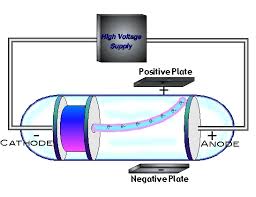
He called the particles “corpuscles,” but they were later renamed electrons. This meant that the ray was negative in charge and that is must consist of particles that have mass. The cathode ray appeared to be repelled by the negative plate and attracted by the positive plate. Thomson tested these ideas by placing negative and positive plates along the sides of the cathode ray tube to see how the cathode ray would be affected. Or was a cathode ray a stream of moving particles? That was the other popular hypothesis. Is it just a ray of energy that travels in waves like a ray of light? That was one popular hypothesis at the time. What is a cathode ray? That’s what Thomson wanted to know. When high-voltage electric current is applied to the end plates, a cathode ray travels from the cathode to the anode. The other electrode is positively charged and known as an anode. One electrode is negatively charged and known as a cathode. It contains a piece of metal called an electrode at each end. A cathode ray tube is a glass tube from which virtually all of the air has been removed. In his research, Thomson passed current through a cathode ray tube, similar to the one seen in the Figure below. This research also led to his discovery of the electron. In 1906, he won the Nobel Prize in physics for his research on how gases conduct electricity. Thomson was born in England and studied at Cambridge University, where he later became a professor. J.) Thomson discovered that there are smaller particles within the atom. It wasn’t until 1897 that a scientist named Joseph John (J. He envisioned them as solid, hard spheres. He thought they were the smallest particles of matter, which could not be broken down into smaller particles. Atoms were discovered around 1800, but almost 100 years went by before electrons were discovered. Electrons are negatively charged fundamental particles inside atoms. But do you know what electricity is? It actually consists of a constant stream of tiny particles called electrons. You probably know that the wires strung between these high towers carry electricity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
